|
Dipole moment can arise in ionic bonds as well as in covalent bonds. Clearly the field points out of the dipole on one end, and into it on the other, following the direction of the dipole moment. A semiempirical calculation of the H2 - H, electric dipole moment has been made by Van Kranendonk and Kiss ( ref. A dipole moment occurs in any system in which there is a separation between the charges. A bond dipole moment is a measure of the polarity of a chemical bond between two atoms in a molecule. We'll start with a simple part of the field – along the axis defined by the dipole moment. The polar molecules have permanent dipoles and nonpolar molecules do not have permanent dipoles. As with the results above, we want our final answer in terms of the dipole moment, not the charge and separation. Dipole Moment Definition: A dipole moment is a measure of the separation of positive and negative electrical charges in a molecule, and is quantified in. Okay, so what do they look like if we look at them from far away? Well, to determine this, we need to look at them as two charges (sigh, again), and look at the field they create at a distance much greater than their separation, \(d\). 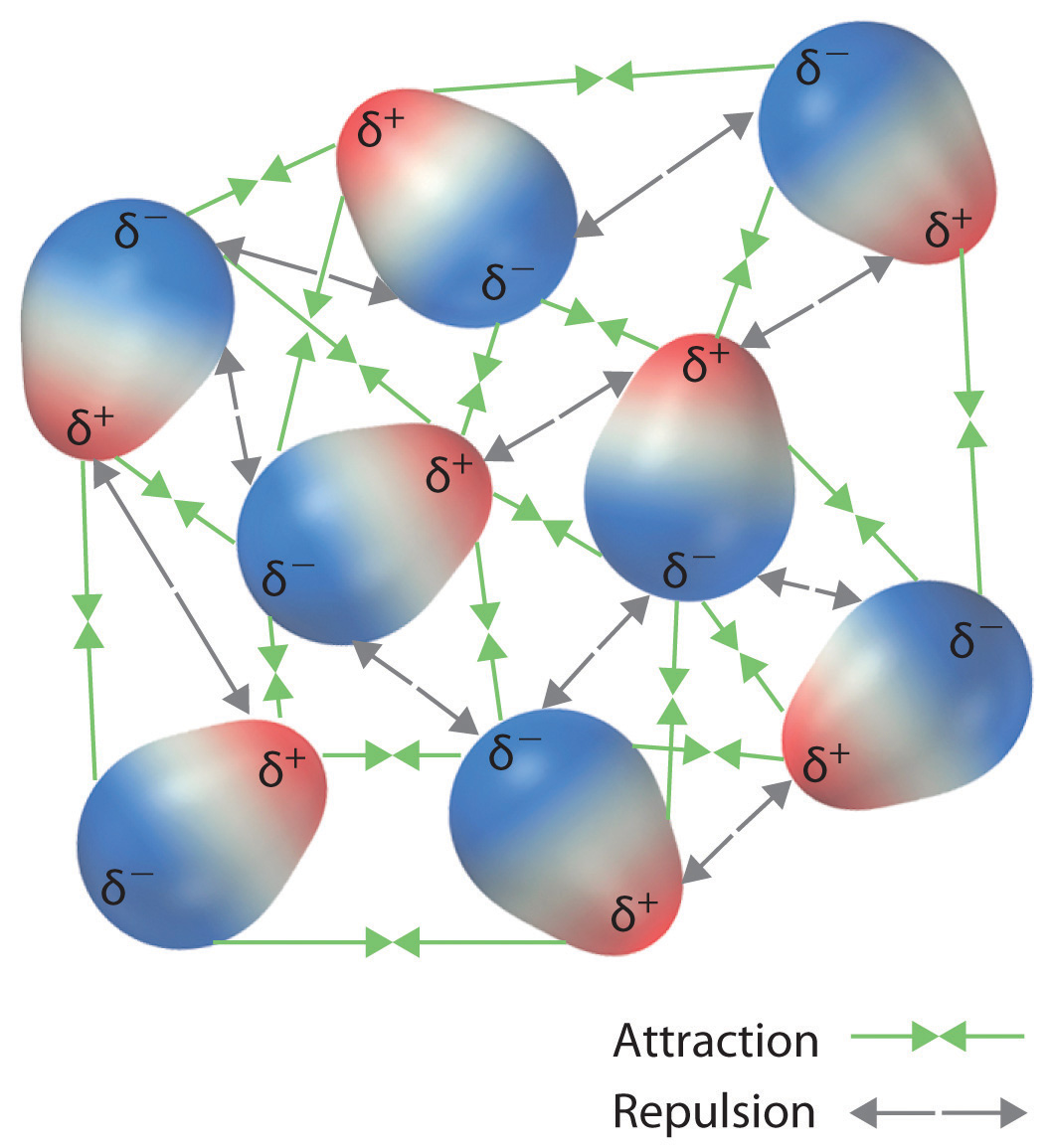
If we are to treat dipoles as "packages," then we have to stop looking at them so closely. The torque vector can now be calculated from the magnetic dipole moment in the same way that the torque exerted on an electric dipole was calculated: We can see that this works for the case shown in Figure 4.2.1: The angle between the magnetic dipole. Note that the energy is a minimum when the dipole moment aligns with the external electric field. For example, the loop in Figure 4.2.1 would have a magnetic moment that points out of the page. = -\overrightarrow p \cdot \overrightarrow E\]
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
